Warning: The binding capacity may also drop significantly if the buffer composition is suboptimal.

It is essential to adjust the pH of the buffer with NaOH after urea addition. Warning: The addition of urea to buffered solutions will cause the pH to drop. Note: Refer to sections Purification Process Optimization and Troubleshooting for technical advice in optimizing the purification results.īuffers recommended in this document are well established examples and can be adapted to achieve optimal conditions for a specific target protein.ĭenature the protein or dissolve the inclusion bodies in a buffer containing 6 M guanidinium-HCl or 8 M urea. Equilibrate the column in a 20% ethanol solution, and store at 2-8 ☌ to prevent cell growth. Warning: If the purification column is not immediately reused, clean the column with 2 column volumes of 2 M imidazole to remove nonspecific binding of proteins.

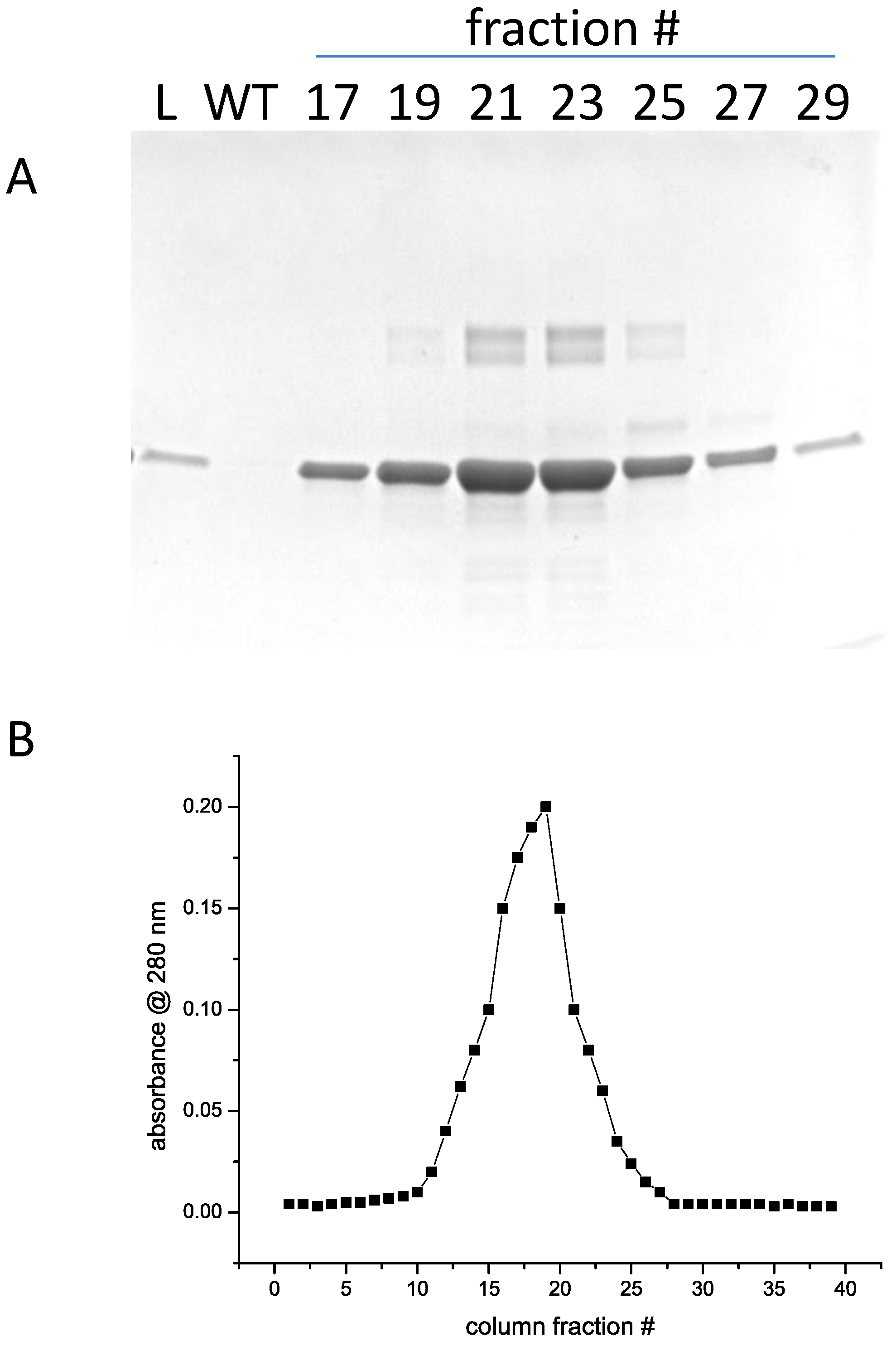
Warning: The amount of imidazole required in the elution buffer for efficient release of the target protein from the resin depends on various parameters: the length of the His-tag, the accessibility of the His-tag. Due to the specific characteristics of cOmplete His-Tag Purification Resin, a protein can already be eluted with approximately 25 mM imidazole. Warning: Protein peaks can be expected between 25 to 45 mM imidazole.
Use high s resin full#
Buffers recommended in this document are well established examples and can be adapted to achieve optimal conditions for a specific target protein.ĬOmplete His-Tag Purification Resin offers full flexibility in selecting the optimal buffer conditions without compromises. Purification of native protein should be performed using optimal buffer conditions for the target protein.
